
MAJOR SECONDARY
ENDPOINT
Week 161,3
66%
(110/167)
vs 11% (9/85) with placebo
P<0.001

ICONIC-TOTAL (N=311): A phase 3, multicenter, randomized, double-blind, placebo-controlled trial that evaluated the efficacy and safety of ICOTYDE 200 mg orally once daily in adult and adolescent patients 12 years and older and weighing at least 40 kg with moderate to severe plaque psoriasis who had a minimum BSA involvement of ≥1% and IGA ≥2 and had failed to respond to ≥1 topical therapies. Patients had at least one of the following baseline conditions: ss-IGA ≥3, sPGA-G ≥3, and/or hf-PGA ≥3. Patients randomized to placebo crossed over to ICOTYDE at Week 16. Efficacy will be assessed through Week 156. Safety will be assessed through Week 160.1,2
View trialICONIC-TOTAL:
Plaque PsO (BSA ≥1%, IGA ≥2) and at least moderate high-impact site involvement.
Primary endpoint was IGA 0/1 and ≥2-grade improvement from baseline:
ICOTYDE 57% (118/208) vs placebo 6% (6/103); P<0.001 (NRI)1,2
Scalp PsO (ss-IGA 0/1) (NRI)
Baseline score ≥3

MAJOR SECONDARY
ENDPOINT
Week 161,3
66%
(110/167)
vs 11% (9/85) with placebo
P<0.001

EXPLORATORY ENDPOINT*
Week 523
72%
(121/167)
49% of patients had completely clear skin
at Week 16 (ss-IGA 0; 82/167)3*
*Exploratory endpoints were not multiplicity controlled. Therefore, statistical significance has not been established.
The same patients may not have responded at each time point.
Week 0
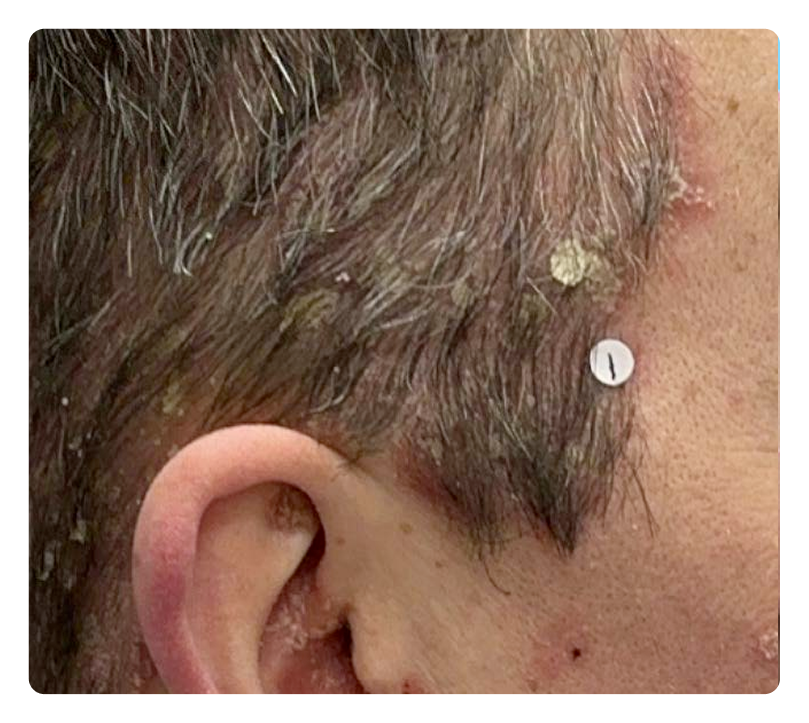
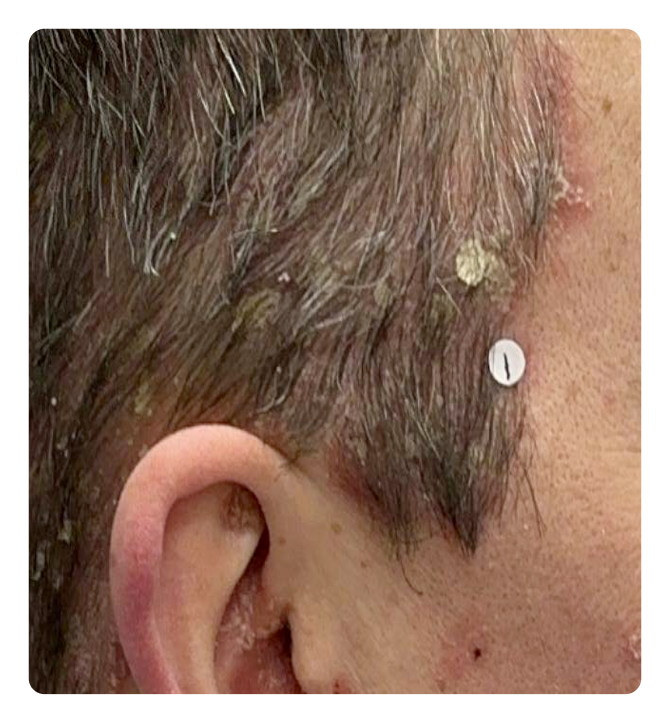
Week 16
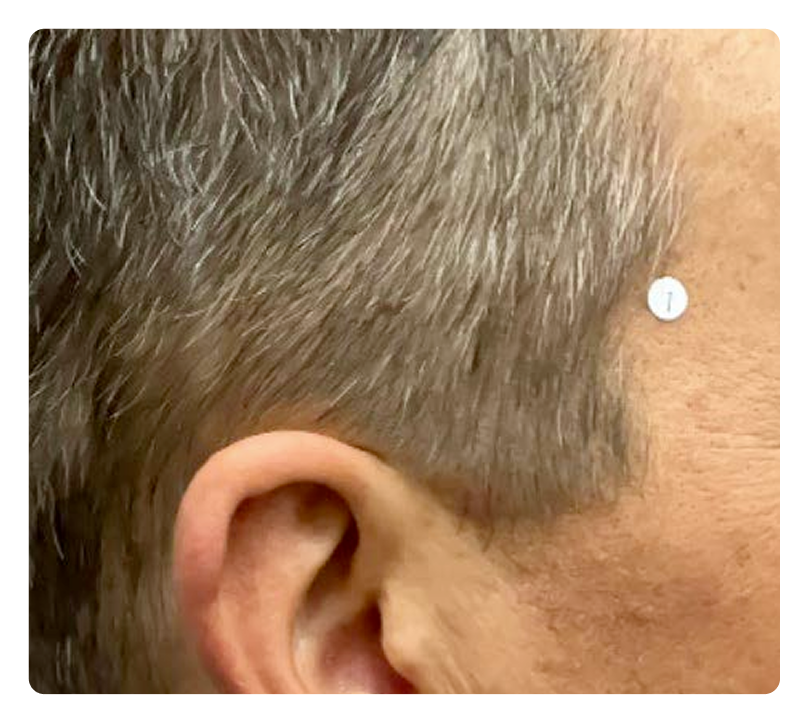
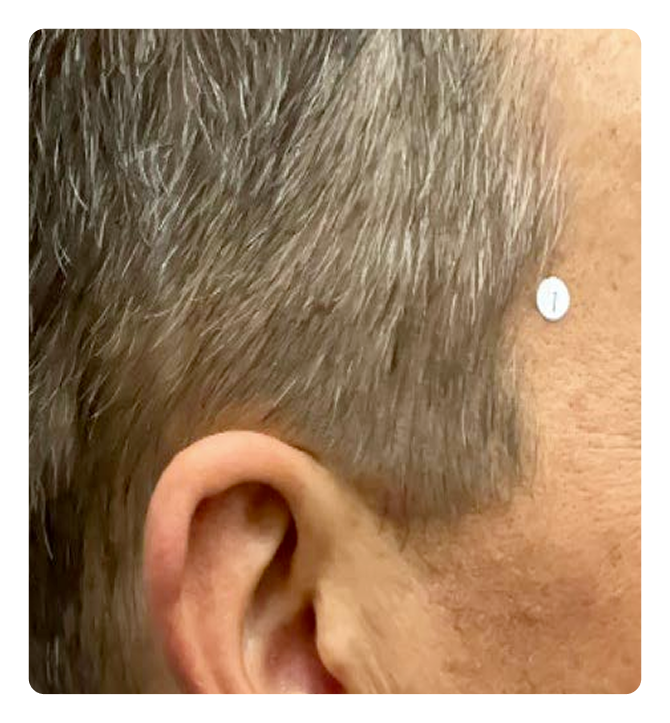
Photos are of a real patient. Images are owned by Janssen Biotech, Inc., a Johnson & Johnson company, from a clinical trial. Individual results may vary.
Genital PsO (sPGA-G 0/1) (NRI)
Baseline score ≥3

MAJOR SECONDARY
ENDPOINT
Week 161,3
77%
(75/98)
vs 21% (9/42) with placebo
P<0.001

EXPLORATORY ENDPOINT*
Week 521,3
85%
(83/98)
62% of patients had completely clear skin
at Week 16 (sPGA-G 0; 61/98)3*
*Exploratory endpoints were not multiplicity controlled. Therefore, statistical significance has not been established.
The same patients may not have responded at each time point.
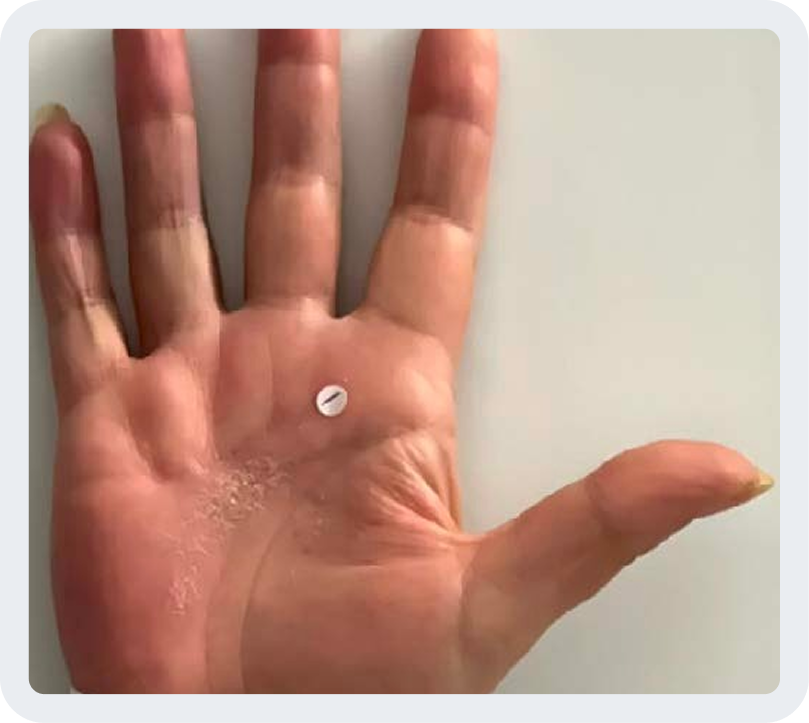
Hand/foot PsO (hf-PGA 0/1) (NRI)
Baseline score ≥3

MAJOR SECONDARY
ENDPOINT
Week 163
42%
(20/48)
P=0.144
Placebo: 26% (6/23)
Not statistically significant.

EXPLORATORY ENDPOINT
Week 523
63%
(30/48)
Data were not multiplicity controlled. Therefore, statistical significance has not been established.
Data were not multiplicity controlled. Therefore, statistical significance has not been established.
Week 16
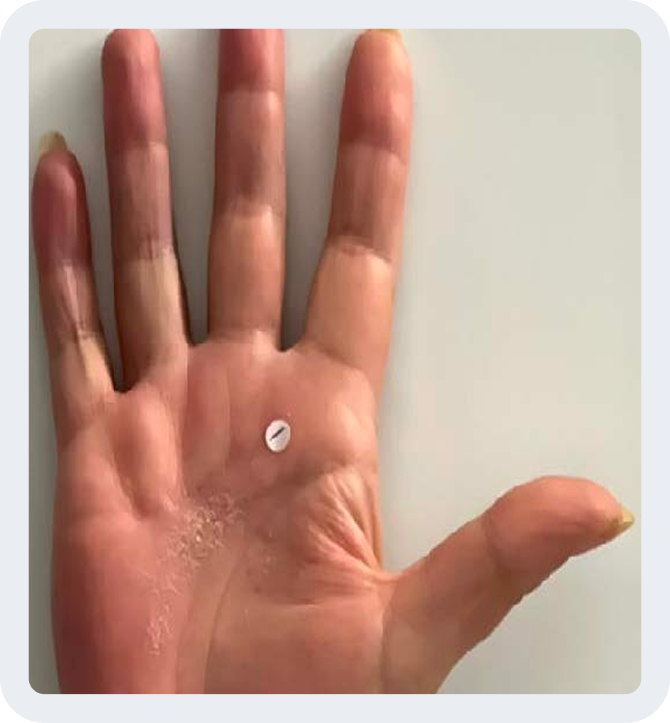
Week 0
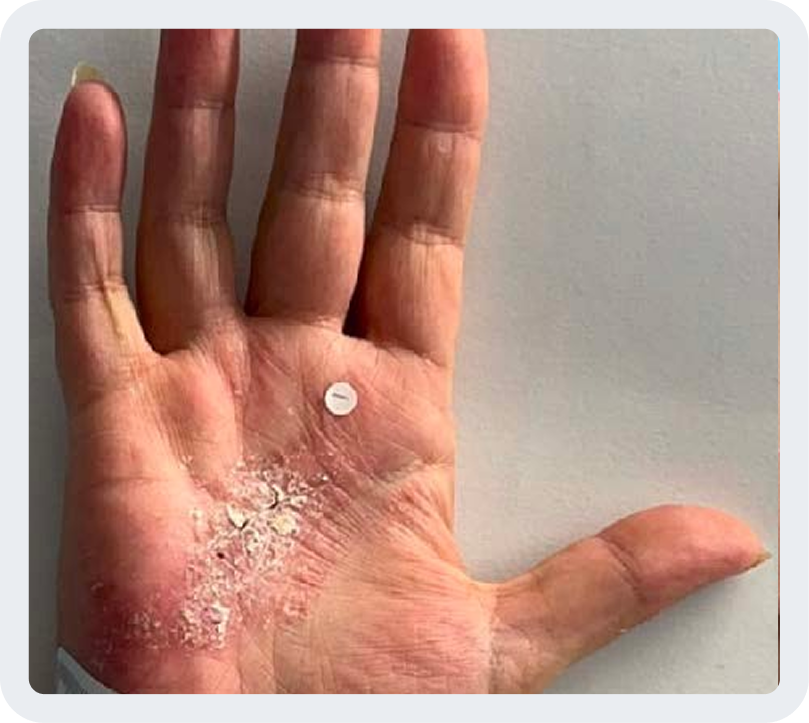
Photos are of a real patient. Images are owned by Janssen Biotech, Inc., a Johnson & Johnson company, from a clinical trial. Individual results may vary.
Itch improvement in high-impact sites at Week 16
Secondary endpoint: ≥4-point improvement from baseline in NRS score1

Scalp itch (NRI)1*
59%
(77/131)
P=0.0081,2*
Placebo: 9% (5/58)

GPSS genital itch (NRI)1†
64%
(44/69)
P=0.0081,2†
Placebo: 13% (4/31)
*Only patients with baseline scores of scalp itch NRS ≥4 and ss-IGA ≥3 were included (ICOTYDE, n=131; placebo, n=58).1
†Only patients with baseline scores of GPSS genital itch NRS ≥4 and sPGA-G ≥3 were included (ICOTYDE, n=69; placebo, n=31).1
EXPLORE SAFETY
PROFILE
Take a closer look at the ICOTYDE safety profile
BSA, body surface area; GPSS, Genital Psoriasis Symptoms Scale; hf-PGA, Physician’s Global Assessment of Hands and Feet; IGA, Investigator's Global Assessment; NRI, nonresponder imputation; NRS, numeric rating scale; PsO, psoriasis; sPGA-G, Static Physician’s Global Assessment of Genitalia; ss-IGA, Scalp-Specific Investigator’s Global Assessment.
References: 1. ICOTYDE [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc. 2. Gooderham M, Lain E, Bissonnette R, et al. Targeted oral peptide icotrokinra for psoriasis involving high-impact sites. NEJM Evid. 2025;4(12):EVIDoa2500155. doi:10.1056/EVIDoa2500155 3. Data on file. Janssen Biotech, Inc.