results in ADOLESCENT PATIENTS
See the data from the ICONIC-LEAD trial

MOST PATIENTS ACHIEVED CLEARER SKIN WITH ICOTYDE vs PLACEBO1,2
ICONIC-ADVANCE 1
Co-primary endpoints at Week 16 (NRI)
ICONIC-ADVANCE 2
Co-primary endpoints at Week 16 (NRI)*
KEY SECONDARY ENDPOINTS:
PASI 100 at Week 24 (NRI)1,2*
*ADVANCE 2 enrolled 731 patients, of whom 723 patients were evaluable for efficacy.
ICONIC-ADVANCE 1 (N=774) and ICONIC-ADVANCE 2 (N=731): Two phase 3, multicenter, randomized, double-blind, placebo-controlled and active comparator–
controlled trials that evaluated the efficacy and safety of ICOTYDE 200 mg orally once daily in adults with moderate to severe plaque psoriasis defined as IGA ≥3, PASI ≥12, and BSA ≥10%. Patients randomized to placebo crossed over to ICOTYDE at Week 16. Patients randomized to deucravacitinib 6 mg orally once daily crossed over to ICOTYDE at Week 24. Patients received open-label ICOTYDE after Week 24 through Week 156. Safety will be assessed through Week 160.1,2
Key secondary endpoints in ADVANCE 1:
IGA 0 at Week 16 and Week 241-3*


IGA 0 in ADVANCE 2 at Week 16: ICOTYDE 37% (118/320) vs placebo 1% (1/82), P<0.001; at Week 24, 40% (128/320) ICOTYDE vs 21% (68/322) active comparator (P<0.001)1,2*†
KEY SECONDARY ENDPOINTS:
IGA 0 at Week 24 (NRI)1,2†
*Multiplicity-adjusted P<0.001, icotrokinra vs deucravacitinib.
†ADVANCE 2 enrolled 731 patients, of which 723 patients were evaluable for efficacy.
CLEARER SKIN WITH ICOTYDE3
Mean PASI improvement from baseline*


ADVANCE 1 other secondary endpoint measured at Week 16. Other endpoints were exploratory.
The same patients may not have responded at each time point.
Data were not multiplicity controlled. Therefore, statistical significance has not been established.
ADVANCE 2: mean PASI improvement from baseline (LS mean)*†
Mean percent improvement values are shown after Week 16 in patients on placebo who switched to ICOTYDE.
*Patients with missing data were not explicitly imputed; they were accounted for in the analysis model.
†ADVANCE 2 enrolled 731 patients, of whom 723 patients were evaluable for efficacy.
Picture this for your patients:
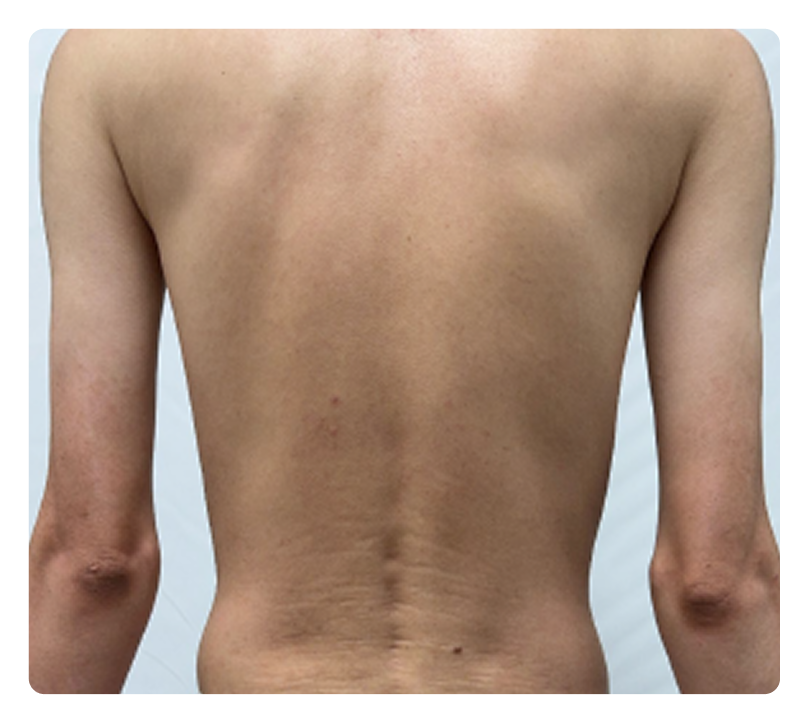
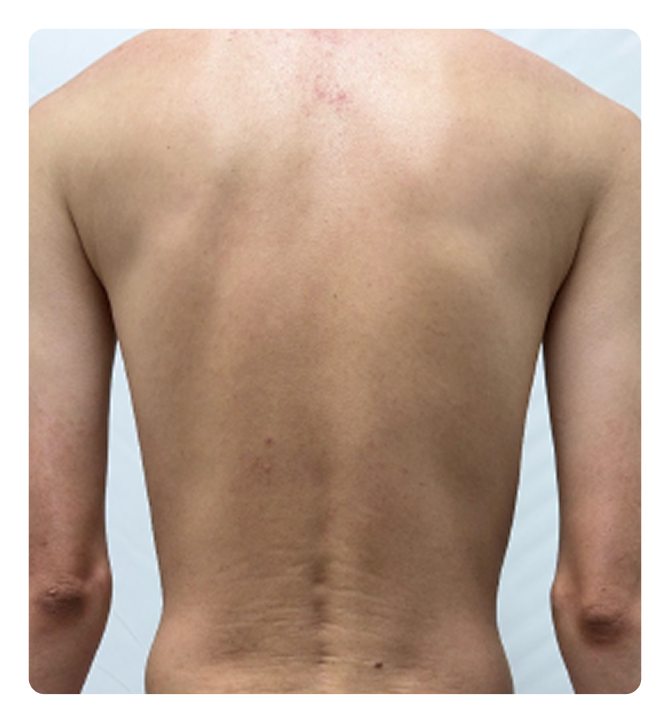
RESULTS FROM ICONIC-ADVANCE 1 and ADVANCE 23
Significant clearance
IGA 0/1; PASI 90
ADVANCE 1
Week 16


Week 0


Complete clearance
IGA 0; PASI 100
ADVANCE 2
Week 16
Week 0
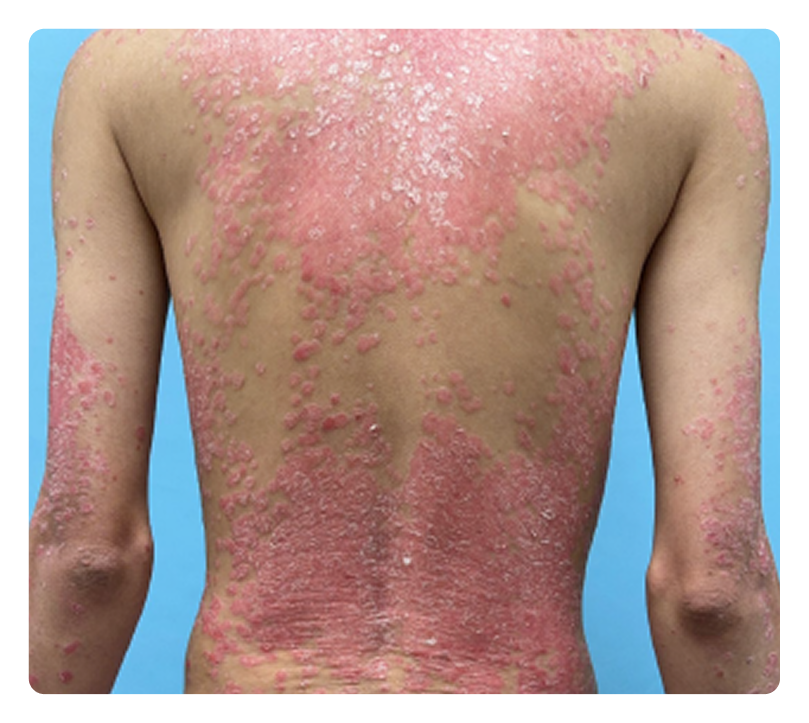
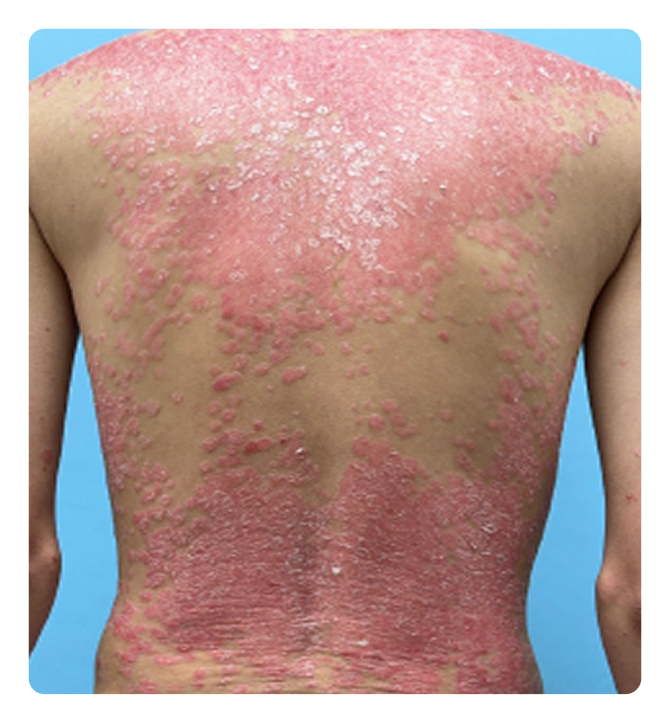
Photos are of real patients. Images are owned by Janssen Biotech, Inc., a Johnson & Johnson company, from clinical trials. Individual results may vary.
results in ADOLESCENT PATIENTS
See the data from the ICONIC-LEAD trial
BSA, body surface area; IGA, Investigator's Global Assessment; LS, least squares; NRI, nonresponder imputation; PASI, Psoriasis Area and Severity Index; PsO, psoriasis.
References: 1. ICOTYDE [Prescribing Information]. Horsham, PA: Janssen Biotech, Inc. 2. Gold LS, Armstrong AW, Bissonnette R, et al. Once-daily oral icotrokinra versus placebo and once-daily oral deucravacitinib in participants with moderate-to-severe plaque psoriasis (ICONIC-ADVANCE 1 & 2): two phase 3, randomised, placebo-controlled and active-comparator-controlled trials. Lancet. 2025;406(10510):1363-1374. doi:10.1016/S0140-6736(25)01576-4 3. Data on file. Janssen Biotech, Inc.